It’s been a swashbuckling tale of biotech adventure. Management and investors in Mesoblast Limited have gone to hell and back in a bold quest to pioneer cell based inflammation therapies. Time and time again the company has announced exhilarating breakthroughs, only to suffer disappointing setbacks. Lately, Mesoblast has taken the biotech drama to the next level. Although this small innovator is not for the faint of heart, the latest twist in the company’s Odyssey may just be the most exciting development yet….
Founded all the way back in 2004, Mesoblast is an Australian company, listed on the Nasdaq stock exchange, that has never gotten a product approved by the FDA. The company is on the cusp of gaining approval in several key indications, but so far, investors have endured 16 years of trials and tribulations before any meaningful revenue has blossomed.
A big part of the reason why the road has been so long and tortuous for Mesoblast is because they insist on doing things the right way. By now it’s very likely you have heard about stem cells, and the enormous therapeutic potential they offer. You may have even heard of people in your community receiving stem cell treatments. Most of the treatments currently offered on a local basis are NOT FDA approved, and have very little scientific evidence behind them. Many of the “solutions” currently offered for large cash payments in orthopedic offices are simply cells that are extracted from your own body, processed in some vague, opaque way, and reinjected right back into you for a large fee. The fact that the materials come from your own body have allowed thousands of unscrupulous practitioners to earn huge profits while ignoring the norms of the scientific community.
While Mesoblast is also trying to offer solutions based on stem cell biology, the similarities to orthopedic chicanery end there. The company describes the science behind it’s offerings:
Mesoblast’s novel allogeneic product candidates are based on rare (approximately 1:100,000 in bone marrow) mesenchymal lineage cells that respond to tissue damage, secreting mediators that promote tissue repair and modulate immune responses.
Mesenchymal lineage cells are collected from the bone marrow of healthy adult donors and proprietary processes are utilized to expand them to a uniform, well characterized, and highly reproducible cell population. This enables manufacturing at industrial scale for commercial purposes. Another key feature of Mesoblast’s cells is they can be administered to patients without the need for donor–recipient matching or recipient immune suppression.
Mesoblast’s approach to product development is to ensure rigorous scientific investigations are performed with well-characterized cell populations in order to understand mechanisms of action for each potential indication. Extensive preclinical translational studies guide clinical trials that are structured to meet stringent safety and efficacy criteria set by international regulatory agencies. All trials are conducted under the continuing review of independent Data Safety Monitoring Boards comprised of independent medical experts and statisticians. These safeguards are intended to ensure the integrity and reproducibility of results, and to ensure that outcomes observed are scientifically reliable.
The company has invested hundreds of millions of dollars and sixteen years striving to produce scientifically accepted, FDA approved products that fight inflation in the body.
During this time, they have produced thousands of patents, and a number of products are almost approved. But it’s been a white knuckle roller coaster ride that would challenge the courage of even the most bold biotech investors.
The Agony and the Ecstasy
The last eighteen months, in particular, have been turbulent for shareholders. On August 14th, 2020, a panel of FDA experts recommended, in a 9-1 vote, approval for Remestemcel-L in pediatric graft vrs host disease. The FDA almost always follows the recommendations of its advisory committees, so the company’s shares tripled in value almost overnight. Investors were excited at the prospect of MESO finally launching its first product after so many years in development.
Shortly after, disaster struck. The FDA chose to ignore the advice of it’s own advisory panel, and require one more safety trial before approval of Remestemcel-L. Although highly frustrating, this is understandable in the context of pediatric care. The FDA is always extra cautious on any novel treatment involving children. An experienced biotech investor could see this development as a temporary setback, but Wall Street was less patient; the shares quickly lost half of their value.
The next gyration in share price came with a hurricane of news surrounding a totally different trial for a totally different indication. On December 15th, 2020, the company released one of the most confusing press releases of the year. Apparently Rexlemestrocel-L, a cardiac related cell product, failed to stop chronic heart failure in a phase III study. However, the cells did seem to dramatically reduce heart attacks and strokes in this very vulnerable patient population. So, here in the same press release, we had a study that indicated both failure and unexpected success. The study design had held out reduction in chronic heart failure as the main endpoint. The medication failed this endpoint. However, a large reduction in heart attacks and strokes was a pleasant, unexpected benefit.
Investors had a hard time knowing what to make of this news. One one hand, it seemed to open up a whole new world of promise for Rexlemestrocel-L (655,000 Americans died of heart disease last year). But on the other hand, it was still a phase III trial that missed it’s primary endpoint. Could this still be Mesoblast’s first approved product? Management vowed to forge ahead:
Mesoblast Chief Medical Officer Dr Fred Grossman said: “We expect the mortality benefit observed in this seminal Phase 3 trial will support a potential path for approval of Rexlemestrocel-L in patients with advanced chronic heart failure. We are planning to meet and discuss potential pathways to approval based on mortality reduction with the United States Food and Drug Administration.”
Once again, Wall Street was unimpressed, and the shares continued to lose value.
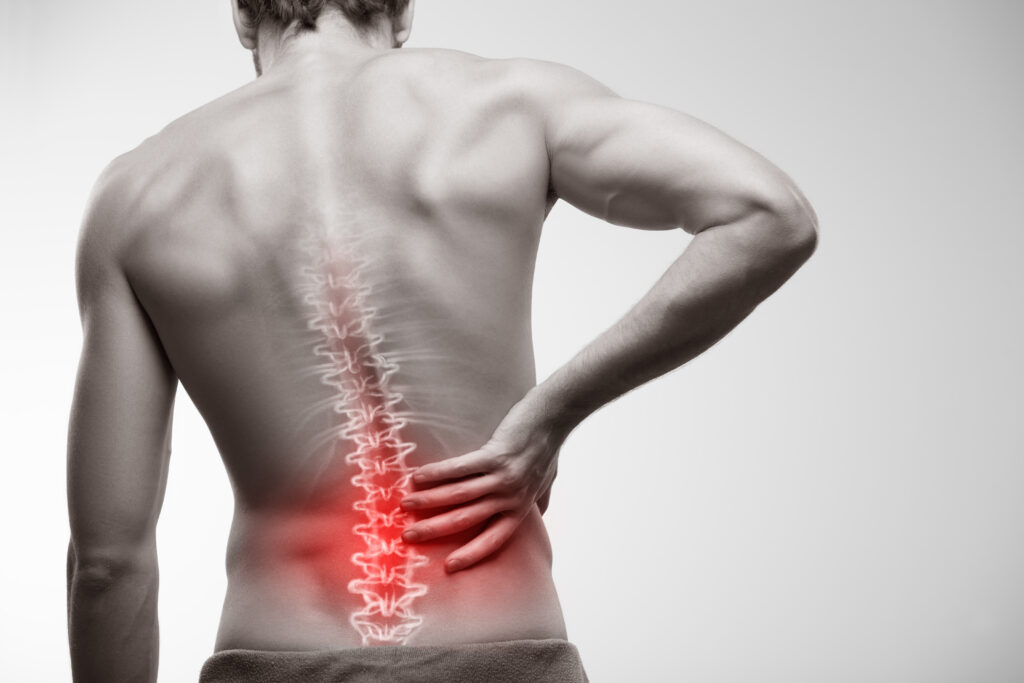
2021 began with even more drama. On February 11th, 2021, the company announced outstanding results in it’s phase III trial related to treatment of back pain. This time $MESO nailed it’s intended endpoint: the company presented definitive data indicating dramatic improvements in pain for patients suffering from degenerative disk disease of the spine. The company was also able to demonstrate significant reductions in opioid use related to its anti-inflammatory cell treatments. By almost any standard, it seemed like Mesoblast had finally discovered the Holy Grail; clear, unambiguous evidence of efficacy against a widespread disease with poor current treatment options.
But Wait, There’s More….
The February announcement was big news to be sure. But $MESO wasn’t done with her surprises. The first surprise was Wall Street’s poor reaction to the good news. Despite the breathless tone of a press release eager to spread good news, the best Wall Street could seem to manage was a “Meh” reaction. The shares barely reacted to the news.
However, shares did react to yet another bombshell that the company released on February 26. In it’s bi-yearly shareholder report, the company casually mentioned that it was running out of cash, and might not be able to continue as a going concern. In the second half of 2020, the company had burned through $60 million, and now had only $70 million left.
In the very same press release, the company announced that it had a new investor lined up. The details were vague. This would be the equivalent of your airplane pilot announcing that the plane is running on fumes, but, not to worry, he knows where the nearest airport is, and he can probably land the plane safely there. Probably. Perhaps the Mesoblast plane would crash and burn, or perhaps not, but the share price certainly continued it’s rapid downward descent.
Just a few days later, in characteristic $MESO style, management banished the gloom and doom with yet another new announcement: Not only had funding been secured, but Mesoblast was about to enter into a very special new partnership:
Chief Executive Dr Silviu Itescu said “We are pleased to receive a strategic investment from the principals of SurgCenter Development, one of the largest private operators of ambulatory surgical centers in the US specializing in spine, orthopaedic and total joint procedures. We expect the deep healthcare knowledge and expertise of this investor group will be of great benefit to the company. The network and infrastructure of surgeons and ambulatory centers operated by SurgCenter may provide unique synergies to facilitate development and market access for Rexlemestrocel, if approved, in patients with chronic lower back pain.” Since its inception in 1993, SurgCenter has partnered in over 230 facilities, with a current portfolio of 80 operational facilities, located in over 20 states throughout the US. SurgCenter is an industry leader in the performance of complex spine surgery and outpatient joint replacement, and has partnered with thousands of surgeons over its three-decade history.
Shazam! Mesoblast strikes again. With the plane’s fuel topped up and some very special new passengers aboard, the company is ready to soar to new heights.
What Matters
If this feels like it is all a lot to digest, it is. The violent swings in this company’s fortunes are enough to give the most experienced investors whiplash.
But there are a few critical points that stand out.
First, Mesoblast now has phase III data that supports the approval of it’s products in three different markets (back pain, cardiac and graft vrs host). At least two of the three potential approvals have vast TAM (Total Addressable Market).
Second, if $MESO can finally get the back pain indication across the goal line, they’re new partnership with Surgcenter Development could mean that sales will rocket out of the gate. These surgery centers tend to cater towards well to do patients who are experiencing a lot of pain. They may well be happy to pay cash for a proven product with FDA approval. If you have ever known anyone suffering from chronic back pain, you will know how bad their current treatment options are. This partnership could be a game changer.
Third, the cardiac data could open up vast new worlds for Mesoblast. They were originally aiming for a very narrow indication (treatment of patients with heart failure). But they wound up with strong evidence that their allogenic stem cells can prevent heart attacks and strokes. This may change the TAM from a relatively narrow, specialized field (Chronic Heart Failure) to something that could help millions of Americans. A bigger TAM means much bigger potential for the company as a whole.
All of that being said, this stock is still very risky. With the new investors coming onboard, $MESO now has just enough cash to get approval for back pain and launch the product. If the company has not achieved it’s long elusive first FDA approval within 18 months, the whole thing could go belly up and the patents could be sold for scrap.
Additionally, Wall Street has become numb to $MESO’s frenetic press releases. The shares actually went down after the latest good news. (SurgCenter drove a hard bargain and bought their shares at a discount from market value, thus diluting current shareholders. Wall Street hates that).
Lastly, the market as a whole is flying high right at the moment. Even the riskiest biotech ventures are drawing record sums of investment. It was relatively easy for Mesoblast to raise funds on a moment’s notice. Many analysts feel that the current market could suffer an epic meltdown, like in 2001. In that scenario, $MESO may not find it so easy to keep the plane from crashing the next time it runs out of gas.
Mesoblast is really a company that exemplifies the classic biotech ethos. It’s innovation or bust for this team. If a wild ride is exciting to you, rather than scary, this may be a company for you. As the saying goes, “No guts, no glory.”
Disclosure: The Sick Economist owns shares in $MESO.




More Stories
2026: THE YEAR THAT PATIENCE PAYS OFF?
WHO WINS IF AGING IS DEAD?
WHY IS AI STRUGGLING TO DISCOVER NEW DRUGS?